HSCT: what to expect
HSCT is a hugely promising treatment for MS. You might also see it called AHSCT. It’s a very intense treatment. This means it comes with risks and there are lots of things to consider.
Read our tips for talking about HSCT with your doctor or nurse
- Do I qualify to get HSCT on the NHS?
- What happens during HSCT?
- What should happen after HSCT treatment?
- How do I get HSCT?
- Getting HSCT and the MS Society
Do I qualify to get HSCT on the NHS?
If you're thinking of HSCT, we recommend you talk to your neurologist about whether it's the right treatment for you. Even if your neurologist thinks it is, it’s not just their decision. If they refer you for HSCT, your case is discussed by a group of experts who would carry out your HSCT. This is called the ‘multidisciplinary team’ (MDT). They look at the risks and benefits for each person, then make a decision. They have the final say.
The NHS have criteria for who can get HSCT. The latest criteria come from the European Society for Blood and Marrow Transplantation (EBMT) and ECTRIMS.
But the NHS and European guidelines don’t match up completely. There might be differences in how guidelines are applied at the centres that offering HSCT. These currently are London, Sheffield and, since late 2022, Cambridge.
EBMT guidance for relapsing MS
HSCT should be offered to people with relapsing MS who:
- have had at least two relapses (or one relapse with signs of new lesions on MRI) in the previous 12 months. This must be despite treatment with one or more of the highly effective DMTs. These are alemtuzumab (Lemtrada), ocrelizumab (Ocrevus), ofatumumab (Kesimpta), cladribine (Mayzent) and natalizumab (Tysabri and Tyruko)
The evidence best supports giving HSCT to people who:
- have an EDSS of 5.5 or less
- are preferably younger than 45 (evidence shows HSCT gets poorer results in people older than this)
- have had MS for less than 10 years
On rare occasions people might also get HSCT if they’ve never had a DMT but their MS is very aggressive from the beginning. They haven’t had MS for long but it’s getting worse very quickly.
EBMT guidance for progressive MS
HSCT may be considered for people with active progressive MS who:
- show signs of active inflammation (relapses and signs of new lesions on an MRI)
This should preferably be in a clinical trial.
NHS criteria for HSCT
The NHS criteria for HSCT in England refer to the EBMT guidance.
The centres that offer HSCT on the NHS follow guidance from the EBMT. This says someone can have HSCT if they haven’t responded to one or more DMTs. The DMTs in question are alemtuzumab (Lemtrada), natalizumab (Tysabri and Tyruko), ocrelizumab (Ocrevus), ofatumumab (Kesimpta) or cladribine (Mavenclad).
If you live in Wales, Scotland or Northern Ireland, you might be able to have the treatment at English hospitals that offer it.
Read the full EBMT guidelines for HSCT on nature website
Read the full NHS England criteria for DMTs, including HSCT (PDF 531KB)
What happens during HSCT?
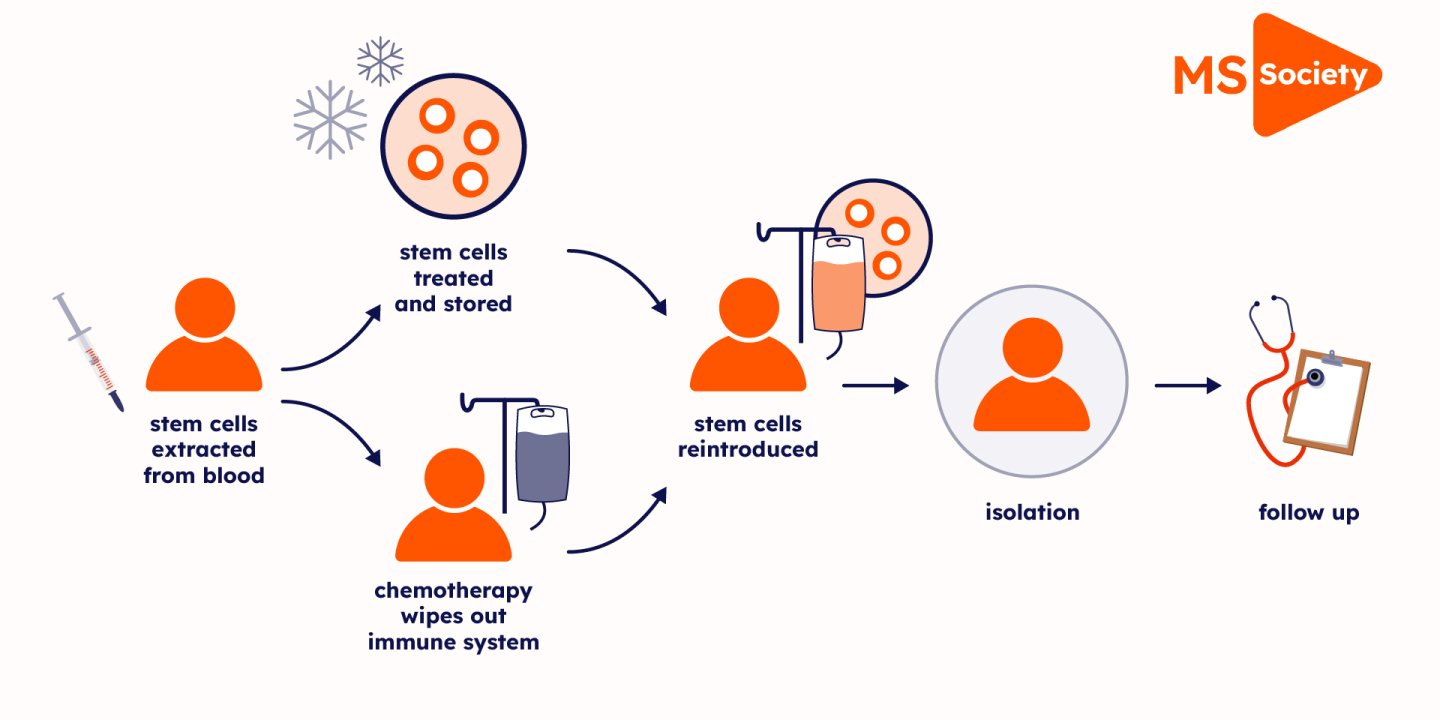
There are several steps to HSCT treatment for MS:
A specialist team reviews your case
Anyone being considered for HSCT will first be reviewed by the specialist team at the treatment centre. This is called the multidisciplinary team (MDT).
If they give the green light for HSCT, they’ll talk to you about fertility issues. The chemotherapy drugs used in HSCT can leave you unable to have children. They can also trigger early menopause.
So, before you start HSCT you might choose to have your eggs frozen in case you one day decide to get pregnant. If you want to father a child, they can freeze your sperm. Talk to your specialist team about how this will be paid for, and how difficult it might be to do.
Drugs get your stem cells ready for collection
The treatment begins with drugs to encourage stem cells to move out of your bone marrow and into your blood. This is known as mobilisation, and allows them to be collected. The drugs include chemotherapy and G-CSF. G-CSF is a man-made version of a protein found naturally in our bodies. You’re given it as injections as an outpatient. The symptoms of MS can get temporarily worse during this stage.
Your stem cells are collected
Around 10 days later, when there are enough stem cells in your blood, they will be removed and stored for later in the procedure. The stem cells are collected through a tube in your arm or neck. Sometimes not enough stem cells are collected. Then you might need another course of G-CSF injections.
Chemotherapy wipes out your immune system
Next, you'll be given an infusion of chemotherapy drugs. These drugs, usually used to treat cancer, wipe out your immune system. This includes the faulty immune cells that are causing your MS. This part of the procedure can take several days and means a stay in hospital.
Chemotherapy may be myeloablative. This means it completely wipes out your immune system. Or it can be a less aggressive version called non-myeloablative. This only partially wipes out your immune system. This version is less hard on the body. In the UK only non-myeloablative chemotherapy is used during HSCT.
The side effects of chemotherapy include nausea and vomiting. You might be given drugs to control this.
Your stem cells are returned to your blood
Finally, your stem cells are transplanted back into your blood by a drip. This usually happens straight away or a day after the chemotherapy. The stem cells help regrow your immune system.
The stem cells get to work
The stem cells start making new blood and immune cells within 10 and 30 days of the transplant. As your immune system isn't working yet, you are more likely to get infections during this period. You're usually put on antibiotics and transfusions to support you. To protect you from infections, you’ll be put in a side room.
Recovery from HSCT
Anyone having HSCT also has to spend around four weeks in hospital in an isolation room while their immune system rebuilds. This isolation can be lonely and challenging. After isolation you can go home but you’re advised to shield for three months. This means staying at home and limiting contact with other people. This is to avoid infections while your immune system is recovering.
Read how three people who've had HSCT dealt with the isolation
HSCT is an intense treatment, so recovery can take some time. Typically, people need between two and six months to recover from HSCT. But for some people, it can take more than a year to fully recover.
What should happen after HSCT treatment?
- The transplant unit should organise regular outpatient monitoring for the first two years following the procedure and afterwards if needed. In some centres this lasts for five years.
- the transplant centre should provide clear guidance on follow up arrangements. This should include the need for urgent advice or treatment for complications if needed.
- The risks of infection persist for long periods after HSCT and antibiotics are recommended for many months, or sometimes even life-long
- The European Bone Marrow Transplant Registry (EBMT) have years of data about how people have got on after having HSCT. This will help your medical team as they check your progress during the months and years after you have HSCT.
How do I get HSCT?
New patients might get HSCT through the NHS. Some are getting it through a clinical trial that is no longer taking new people. Some people go private and pay for HSCT at UK centres, or they go abroad for it. We’re working to help people with MS get access to the right treatment for them.
Through the NHS
HSCT is available on the NHS. At the moment, it’s only considered for people who meet very specific medical criteria.
We know the following UK hospitals offer HSCT for MS outside of a trial. The Royal Hallamshire Hospital in Sheffield, one in Cambridge and several in London: King’s College, Imperial College, St George’s Hospital, University College London Hospitals and Barts Health Trust.
You can be referred to these hospitals from other hospitals in England. People in Wales, Scotland and Northern Ireland who qualify to get HSCT might be able to have the treatment at the hospitals in England.
The criteria might vary slightly between each hospital. So it’s worth checking with your neurologist about which place would suit you best.
Going private in the UK
You can have HSCT privately at the centres in Sheffield and London. When it comes to who qualifies for HSCT, there can be different rules between the centres and compared to the NHS.
Going abroad
If you don’t qualify for HSCT on the NHS, it’s possible to get the treatment abroad. This option is very expensive. HSCT is available privately in several countries including Mexico, Russia, Israel and India.
It’s important to check the safety record of any centre. The EBMT have safety standards which they recommend for HSCT centres.
We'd urge anyone thinking of going abroad for treatment to first talk their options through with their MS nurse or neurologist.
Read more about having HSCT outside the NHS
Getting HSCT and the MS Society
We’re working alongside clinicians to help make sure people with MS can get treatments that are right for them at the right time. This includes HSCT for people who are eligible.
We advocated for HSCT to be available on the NHS. We were part of an NHS England working group that developed treatment guidance which made HSCT available for the NHS in England for the first time. It's unusual for an unlicensed treatment to be made available and we were pleased to play a part in making this happen.
We played a key role in getting the people together who developed the new phase III clinical trial, Star-MS. This trial is comparing HSCT with four other highly effective disease modifying therapies (DMTs). Recruitment to the trial ended in September 2024, so they’re no longer looking for people to take part. Everyone stays part of the trial for two years. We should hear the results by 2027.
In Wales, Scotland and Northern Ireland we’re aware of a small number of people are being referred for HSCT in England, but access remains very limited.
Last full review: 1 August 2025
Next review date: 1 August 2028
We also update when we know about important changes.